The principle of life
When we look at creatures with the philosophy of
thermodynamics , we are surprised, that there
isn't any kind of chaos, which the second law of
thermodynamics predict. If
we ask an expert of thermodynamics about this paradox, he would
explain, that thermodynamic laws are made for closed systems and living
creatures aren' t closed. This is a very good argument, but it doesn't
explain, why such a high order exist. This
article describes
it by the concept of irreversibility .
Chemical processes
in living creatures are determined by
enzymatic reactions. If
we consider enzymes as normal
catalysts, then also exist the dogma, that catalysts can't shift the
position of a chemical equilibrium, because both directions are speeded
up. However this assumption can't be correct in
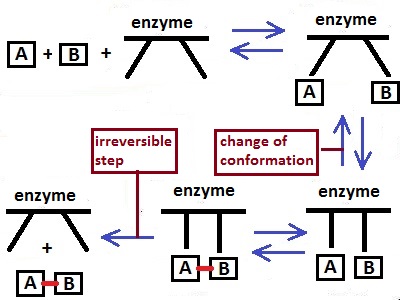
a complex reaction mechanism, like in the
upper picture.This enzyme
catalyse the synthesis
of AB molecules. It changes his conformation during a complex
reaction
mechanism and connect the A and B to
AB. But the last step is an irreversible one,
because for a back reaction, the AB molecule can connect
only with one arm of the enzyme.
If an enzyme
speed up only one direction, it also can shift
the position of the
equilibrium in kinetic reasons .
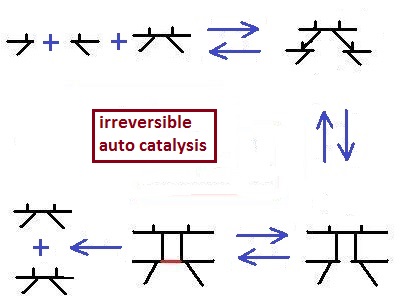
The same principle is
possible in an auto catalysis, in
which the enzyme produce itself .
This
we can consider as a prinziple of live. Also here
we have "chemical reproduce" in which the enzyme produce more
"descendants" than thermodynamic calculations allow.
Searching in living cells for such irreversible
processes, we can discuss complex mechanisms like the protein
biosynthesis or the aspiratory chain and photosynthesis.
back
next :
Irreversibility and the fhoen wind
Link to: Newtons bucket experiment
E-Mail:
newtonexperiment@web.de
<